
In modern glass melting, alkali vapor corrosion has become one of the most underestimated drivers of premature lining wear, unplanned shutdowns, and glass defect risks (stones, cords, seeds, and composition drift). This tutorial-style guide explains how different refractory families behave under high-alkali atmospheres, why high-purity β bauxite blocks are increasingly specified in critical zones, and what installation/maintenance details determine whether the lining reaches its campaign target.
Alkali species (typically Na/K compounds) volatilize from batch and melt, then circulate in the superstructure and crown space. In oxygen-enriched and high-pull furnaces, operators often observe faster deposition and chemical attack because the atmosphere stays hot and reactive for longer residence time. The damage pattern is rarely uniform: it concentrates where alkali vapor meets temperature gradients and where condensation/reevaporation cycles repeatedly occur.
Industry operating data commonly shows that in alkali-intensive production (e.g., container glass with high cullet and soda), superstructure refractory wear rates can accelerate by 25–60% compared with lower-alkali campaigns, especially near hot spots and burner ports.
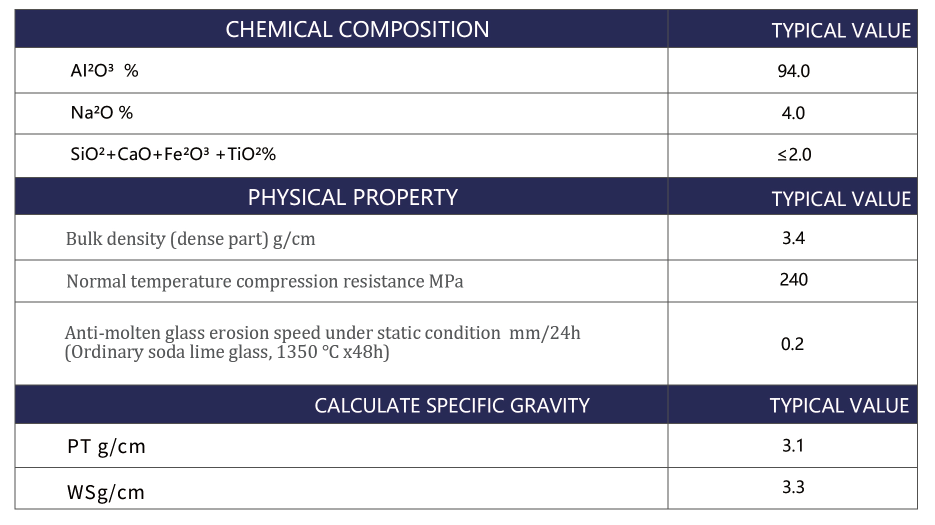
In strong alkali vapor conditions, “high refractoriness” alone is not enough. Procurement teams and furnace designers typically evaluate a shortlist of properties that directly correlate with campaign life and glass quality:
| Property | Why it matters (strong alkali vapor) | Practical target (reference) |
|---|---|---|
| Chemical purity & phase stability | Reduces reactive impurities that form low-melting phases with Na/K vapors | High Al2O3 with low Fe2O3/TiO2 and controlled alkalis |
| Apparent porosity & permeability | Lower penetration of condensates and fewer corrosion pathways | Often < 18–20% for dense blocks (application-dependent) |
| Thermal shock resistance | Withstands temperature gradients at ports/crown without cracking | Stable microstructure, low microcracking after cycling |
| Hot strength & creep resistance | Maintains geometry; reduces joint opening and vapor ingress | High load-bearing stability in 1200–1500°C superstructure ranges |
| Low contamination risk | Minimizes stones/cords from corrosion fragments and reaction products | Low spalling tendency; controlled grain size distribution |
Note: Specific acceptance limits should be aligned with furnace zone (crown, breast wall, doghouse, port neck, regenerator) and the plant’s batch chemistry. In practice, selecting a “best overall” refractory is less effective than building a zone-by-zone alkali exposure map.
Below is a simplified comparison of common refractory families used in glass furnaces, focusing on their behavior under strong alkali vapor. The intent is not to “rank” materials universally, but to clarify trade-offs that influence corrosion rate and defect risk.
| Refractory type | Strengths in alkali vapor | Typical limitations | Best-fit zones (examples) |
|---|---|---|---|
| Silica (superstructure-grade) | High temperature capability; proven crown solutions in many furnaces | Sensitive to alkali attack and condensation cycling; risk of reaction layer and spalling if alkali load is high | Crown (when alkali load is controlled), areas with stable thermal profile |
| High-alumina bricks | Improved alkali resistance vs. silica; good hot strength in many designs | Quality varies widely by raw material purity and porosity; may still form low-melting phases with severe alkali | Breast wall, superstructure transitions, burner surrounds (design-dependent) |
| High-purity β bauxite blocks | Excellent resistance to strong alkali vapor; stable microstructure; helps reduce reaction products that can contaminate glass | Requires correct joint design and controlled installation to fully realize corrosion resistance | Hot-face superstructure, alkali hot spots, port-adjacent structures, areas where vapor deposition is heavy |
| AZS / fused-cast materials | Strong corrosion resistance in glass-contact applications; proven for aggressive melts | Not a universal solution for vapor-phase alkali issues in superstructure; design and compatibility are critical | Glass-contact zones (tank blocks, throat, sidewall—per specification) |
The practical trend in high-alkali operations is a more deliberate split: materials optimized for glass-contact corrosion are not automatically the best for alkali vapor corrosion. That is where dense, high-purity alumina-based solutions such as β bauxite blocks are gaining attention in superstructure selection.
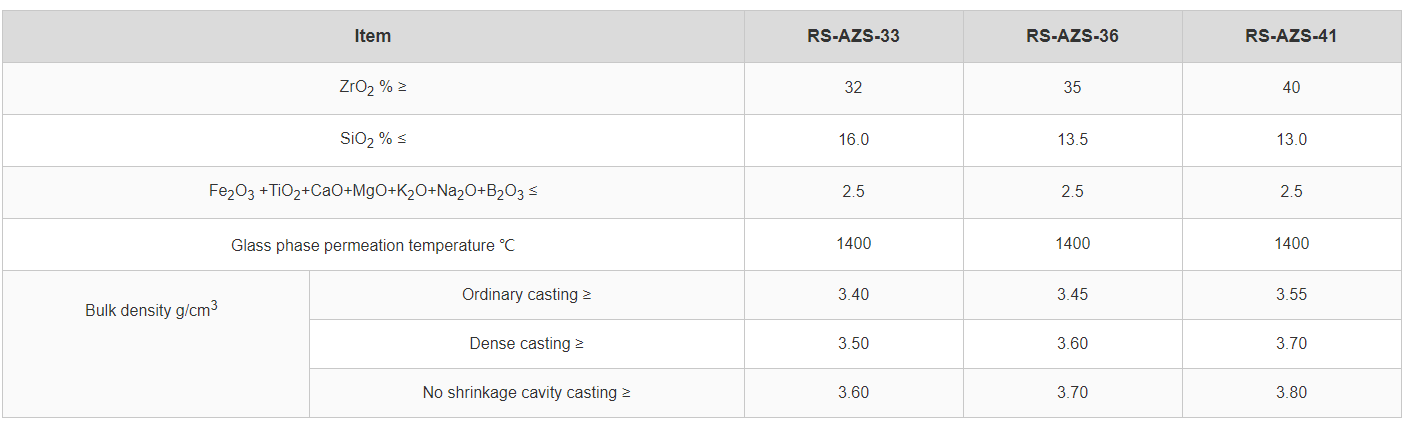
β bauxite blocks (when produced from carefully selected, high-purity bauxite and processed for density and phase control) are specified for one straightforward reason: they slow down the chemical pathway that turns alkali vapor into a destructive, low-melting reaction layer.
Reference engineering range (for evaluation only): In high-alkali superstructure scenarios, plants often aim for dense blocks with < 18–20% apparent porosity and maintain tight joint control to limit vapor ingress. Actual targets must match your furnace design and thermal profile.
Alkali corrosion is not only a maintenance cost—it is a quality risk. When reaction layers grow and then flake off, fragments can travel into the melt or fall into sensitive areas, increasing stone-related defects. Using more alkali-resistant blocks in high-deposition zones can reduce the frequency of corrosive spalling events and stabilize the furnace atmosphere chemistry at the hot face.
“Vapor-phase alkali is a campaign-life multiplier: when the superstructure resists deposition-driven corrosion, geometry stays stable longer, joints stay tighter, and the furnace becomes easier to control.” — Field note from a glass furnace rebuild review
A reliable selection process starts with mapping the alkali vapor load—not guessing it. Many engineering teams now treat alkali corrosion like a “gas-side slag”: it has a chemistry, a transport path, and a condensation pattern.
If the dominant failure is deep penetration and reaction layer growth, prioritize dense, low-permeability materials (often where high-purity β bauxite blocks deliver value). If the dominant failure is thermal cycling and cracking, evaluate thermal shock resistance and joint engineering with equal seriousness.
For procurement clarity and supplier accountability, include a short list of measurable criteria in RFQs: chemistry (major oxides), apparent porosity, bulk density, cold crushing strength, and thermal expansion behavior. Plants that tightened specifications and inspection often report lower early-life cracking incidents and more predictable hot-face stability during ramp-up.

Even premium refractories can underperform if the furnace is assembled in a way that invites alkali vapor into joints and interfaces. The objective is simple: keep the hot-face geometry stable and the infiltration pathways minimal.
Deposition pattern changes
A shift in deposits often signals atmospheric change or a new hot spot that accelerates corrosion.
Joint opening / seal failure
Small openings can become major vapor inlets under draft and burner momentum.
Early spall signatures
Fine cracks or local flaking at the hot face can precede defect events.
A container glass producer running high cullet rates observed accelerated wear near port-adjacent superstructure areas. The maintenance team reported frequent deposit build-up and periodic hot-face flaking that correlated with spikes in stone-related defects. After a rebuild review, the plant executed a targeted upgrade: critical hot-spot zones were redesigned for tighter joints and equipped with dense, high-purity β bauxite blocks to improve alkali vapor resistance.
These results are strongly dependent on furnace design, batch chemistry, firing method, and heat-up/operation discipline. However, the pattern is consistent with broader field experience: alkali-resistant superstructure materials + tight installation control tends to deliver a measurable stability gain.
A practical approach is to combine (1) visual signs of deposit-driven reaction layers, (2) location correlation (ports/crown shadow zones), and (3) sample analysis of deposits and hot-face layers for Na/K enrichment. If cracking dominates without significant reaction layers, thermal cycling may be primary; if reaction layers and glazing dominate, alkali vapor is often central.
Not necessarily. They are commonly evaluated for hot-face superstructure areas where alkali vapor load is high—such as port-adjacent structures, transition zones, and areas with persistent deposition. The best placement depends on your alkali route map and thermal gradients.
Typically: chemical composition (major oxides), apparent porosity, bulk density, cold crushing strength, refractoriness under load (or equivalent hot performance indicator), and dimensional tolerances. For alkali-focused projects, request any available alkali vapor corrosion test references and past furnace application records.
Inconsistent joints and unsealed gaps. Alkali vapor exploits the smallest pathway, and once deposits form inside joints, corrosion accelerates. Tight workmanship, compatible jointing materials, and a disciplined heat-up program are often as important as the block grade itself.
For glass producers facing persistent deposition, corrosion flakes, or early superstructure wear, Zhengzhou Rongsheng Refractory offers high-purity β bauxite blocks engineered for demanding alkali vapor environments—supporting lining stability and helping reduce glass contamination risk in critical zones.
Share your furnace type, pull rate, fuel/oxy configuration, and any deposit analysis data. Rongsheng’s team can suggest suitable block grades and placement strategy for high-alkali vapor hot spots.
Get Rongsheng High-Purity β Bauxite Blocks Technical SupportTypical response includes: product datasheet, recommended zones, and installation checkpoints for alkali vapor resistance.










